In these days, the energy storage systems are playing an increasingly important role in different fields, and the relatively typical fields are like electric vehicles, power systems and some other fields. In this context, the super capacitors, as an energy storage technology, possesses excellent performances such as high power density, maintenance-free, and long life, and it have become the focus of attention in academia and industry.
This section mainly will introduce the electrochemical mechanism of supercapacitors. The energy storage in supercapacitors is mainly in the interface between electrodes and electrolytes. This energy storage method has a great relationship with the electrode materials used. When the two electrodes of a supercapacitor are made from different types of materials, in this case, a comprehensive analysis of the energy storage mechanism of the product will not fully understand the working principle of the super capacitor. Based on this point, this section will briefly introduce the working principle of the super capacitor first; then elaborate the energy storage mechanism of different electrode-electrolyte interfaces, classify supercapacitors according to different electrodes and electrolytes, and introduce some electrical performance characteristics of supercapacitors.
As shown in Figure 1, the construction of the supercapacitor mainly consists of several key components, including current collectors, electrodes, electrolytes, and separators. The role of the separator has the same function as the separator in the battery. It isolates the two electrodes to prevent short circuit between the electrodes and allows ions to pass through. The basic principle of supercapacitor energy storage is to store electrical energy through the electric double-layer capacitance formed by the charge separation on the interface between the electrolyte and the bath solution.
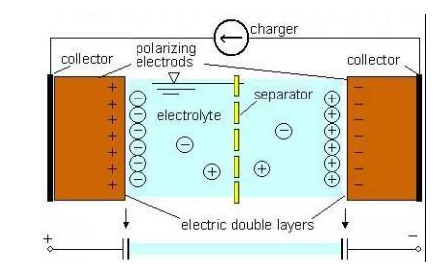
Figure 1: Schematic diagram of supercapacitor structure and working principle
There are many materials used in the manufacture and production of supercapacitor electrodes and electrolytes. In order to deeply understand the energy storage mechanism of supercapacitors and optimize the performance of supercapacitors, it is usually necessary to use two experiments, the cyclic voltammetry curve, and the constant current discharge to characterize the electrode performance of the different supercapacitor. Figure 2 shows two experiments, the cyclic voltammetry curve, and the constant current discharge of the supercapacitor, under different energy storage mechanisms, where a and c represent the cyclic voltammetry curve and the constant current discharge curve of the supercapacitor electrode, under the storage mechanism of electric double layer capacitance and pseudo capacitance, respectively; b and d respectively represent the cyclic voltammetry curve and constant current discharge curve of the supercapacitor electrode under the Faraday capacitor storage mechanism.
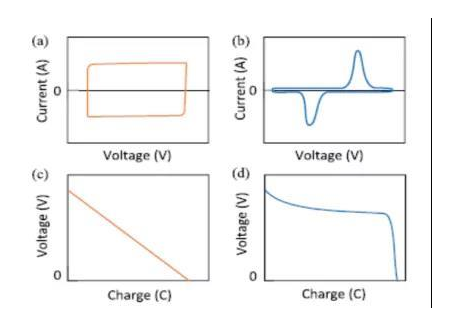
Figure 2: Cyclic voltammetry curves and constant current discharge curves of electric double layer capacitors under different storage mechanisms
1. The storage mechanism of the electric double layer capacitor
The electric double layer effect is that the positive and negative charges should be separative, which are generated by the accumulation at the electrode-electrolyte interface. It is the main mechanism for energy storage of supercapacitors such as activated carbon, carbon fiber, carbon felt and other carbon materials. The formation of the electric double layer effect is mainly caused by the increase or decrease of high-energy conduction band electrons on the electrode surface, which causes the movement of positive and negative charges in the electrolyte solution on the interface side, which is used to balance the charge imbalance caused by the change of the high-energy conduction band electrons on the electrode surface.
Considering the charge density on the electrode surface is depended by the applied voltage, the electric double layer capacitance differs depending on the voltage. The electrochemical reaction in the electric double layer capacitor mainly occurs on the electrode surface, and it is usually the adsorption and desorption behavior of anions and cations. The cyclic voltammetry curve of the electric double layer capacitor has a rectangular shape as shown in Fig. 2 (a), and the constant current discharge curve of this type of material has a linear relationship, as shown in Fig. 2 (c).
The electric double layer effect occurs at the interface between the electronic conductor and the ionic conductor, which is present in almost all electrochemical energy storage systems. However, it is generally regarded as a side reaction in electrolyzers, fuel cells, and batteries, and it will not be regarded as the main energy storage mechanism. On the contrary, the working principle of supercapacitors is based on this effect, which requires supercapacitors to maximize this effect in the design and development process.
2. The pseudocapacitance storage mechanism
The pseudocapacitance, also known as Faraday pseudocapacitance, are the capacitance that related to the electrode charging potential, when it is two-dimensional or quasi-two-dimensional spaces in the electrode surface or bulk phase, and when the electroactive substances undergo underpotential deposition, and highly reversible chemical adsorption, desorption or oxidation, and reduction reactions occur. It is the main mechanism for energy storage of metal oxides, metal carbides, and conductive polymer supercapacitors. Although these reactions are very similar to those in batteries, because both their charges pass through the electric double layer capacitor, the difference is that the formation of pseudocapacitance is more like caused by special thermodynamic behavior. The cyclic voltammetry curve and constant current discharge curve of the pseudocapacitance are similar to the electric double layer capacitor. Unlike the electric double layer capacitor, the pseudo-capacitor has a higher energy density, but is limited by the electrochemical reaction kinetics and the irreversibility of the reaction. As a result, the charge and discharge power and cycle life of the pseudo capacitor are smaller than the electric double-layer capacitor. It should be pointed out that due to the presence of active functional groups, most supercapacitor electrodes have pseudocapacitance. For example, the electrochemical response of electric double layer capacitors composed of nanomaterials such as graphene is mainly formed by redox reaction caused by defects in carbon materials.
3. The Faraday reaction storage mechanism
This storage mechanism is mainly based on the redox reaction of metal cations in the electrode, usually accompanied by the redox reaction of metal cations. The extraction and insertion of metal cations in the electrode material phase extraction will cause the gain and loss of electrons in the material and then stores energy. It mainly includes two ways of material phase transformation or alloying reaction. When these electrodes are charged and discharged, a plateau voltage will appear, which corresponds to the redox peak voltage in the cyclic voltammogram, as shown in Figure 2 (b) and 2 (d). Compared with the other two types of capacitors, Faraday capacitance have higher stored energy, which is generally 10-100 times that of electric double layer capacitors.
Some electrode materials that exhibit Faraday effect, such as Ni (OH) 2 or similar battery electrode materials, are considered to be pseudocapacitive materials in many literatures, which is confusing to the readers. Although this type of material has a higher energy storage energy density and is limited by the solid phase diffusion of the material ions, the high-power charge-discharge performance is far worse than the pseudocapacitance material.
There are many classification standards for the supercapacitors. This article will mainly introduce two classification methods. The first one will be classified according to the different energy storage mechanisms of the electrode materials, and the second one will be classified according to the different electrolytes.
1. Classification according to different energy storage mechanisms
According to different energy storage mechanisms, supercapacitors can be divided into symmetric supercapacitors, asymmetric supercapacitors, and hybrid supercapacitors.
2. Classification according to different electrolytes
According to the electrolyte type, it can be conventionally divided into aqueous electrolytes and organic electrolytes. Among them, aqueous electrolytes include: 1. The acidic electrolytes, mostly using 36% H2SO4 aqueous solution as electrolytes. 2. the alkaline electrolytes, usually using strong bases such as KOH and NaOH as electrolyte, and water as solvent. 3. the neutral electrolyte, usually using KCl, NaCl and other salts as electrolyte, and water as solvent, mostly used for electrolyte of manganese oxide electrode material; the organic electrolyte usually uses lithium salt represented by LiClO4, TEABF4 as typical representative quaternary amine salts are used, the organic solvents usually use PC, ACN, GBL, THL and so on, and the electrolyte is close to saturation solubility in the solvent. It also includes solid electrolytes. With the continuous breakthrough of solid-state electrolytes in lithium-ion batteries, such electrolytes have become a research hotspot in the field of supercapacitor electrolytes.
This section will briefly discuss the electrical performance of supercapacitors. It is hoped that analysis will reveal the causes of some special phenomena of supercapacitors and analyze the impact of these phenomena on the performance of capacitors. The following will also discuss how to choose the supercapacitor that matching different situation correctly, based on the requirements of different supercapacitor applications fields.
The variable capacity is one of the characteristics of supercapacitors, although this feature is not the most critical performance of supercapacitors. But when SC is part of the energy system, it needs to be considered. This is because the capacity variation of the supercapacitor in the entire voltage range is between 15% and 20% of the rated capacity, which cannot be ignored in most designs of energy systems. The capacitance of the supercapacitor can be measured by formula (1). The formula associates the charge and the voltage stored between the electric double layers, which indicates that the amount of charge stored between the electric double layers is proportional to the voltage. As the voltage increases, the charge distribution density near the electric double layer will become higher.
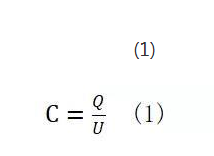
U is voltage and Q is charge
In addition to the influence of voltage on the capacitance of the supercapacitor, the ambient temperature also affects the capacitance of the supercapacitor. Although supercapacitors have a wide operating temperature range, wide temperature changes will have a certain impact on the capacitance of supercapacitors. As a part of energy storage tools, when designing the system, the impact of ambient temperature on the capacitance of supercapacitors should be fully considered. Temperature mainly affects the capacitance of supercapacitors by affecting the anion and cation Brownian motion on both sides of the electric double layer. Generally speaking, temperature affects different ion Brownian motions differently, which means that when the temperature increases, different anion and cation Brownian motion speeds increase. A larger gap will reduce the capacitor capacitance. Some studies have reported that when the temperature changes by 1°C, the capacitance of the supercapacitor will change by 0.1%, indicating that when the operating temperature of the supercapacitor changes by 80 ° C, there will be an 8% capacity change. Although the temperature change has little effect on the capacitance of the supercapacitor, it should be paid enough attention in the system design.
Usually, the supercapacitor has considered the matching performance between the size of the electrode pore and the size of the electrolyte in the design process. It should be pointed out that the supercapacitor electrolyte solvent is usually composed of polar molecules, such as water molecules, acetonitrile, etc. These polar molecules will solvate the ions and combine into larger and more stable units. When the diameter of the electrode pore is smaller than the diameter of the free ion, solvated ions and free ions will not pass through the hole and will not affect the electric double layer capacitance; when the pore is larger than the solvated ion, the solvated ion will pass through the hole; When the pores are between the diameter of ions and solvated ions, free ions will pass through the pores, and the solvated ions will be desolvated to form free ions into the pores.
This process requires energy. The existence of the latter two holes will affect the electric double layer charge distribution. The charge distribution has a certain relationship with the fast rate of charging and discharging and the life of the capacitor.
The study found that it is precisely due to the existence of this solvation phenomenon that the supercapacitor cannot achieve full discharge under high power. It is reported that depolarized ions and dissolved polarized ions account for about 20% of the electrode surface area in commonly used capacitors.
The study has found that it is the existence of this solvation phenomenon that enable the supercapacitor to achieve full discharge under high power. It is reported that depolarized ions and dissolved polarized ions account for about 20% of the electrode surface area in commonly used capacitors.
During charging and discharging, the ions and the electrons in the supercapacitor will have some movements. On the one hand, due to the Joule heat generated by the movement of electrons, it will convert the kinetic energy of the electrons into heat energy, which will dissipate through the conductor. On the other hand, the ions will be rubbed with other ions during the movement of the electrolyte to generate heat. The voltage which offsets these two energy consumptions is proportional to the transport ion or current, and this phenomenon is called the ohmic polarization phenomenon of supercapacitors.
Generally, the effect of temperature change on electrons and ions is opposite. For solid-state electrodes, when the temperature increases, the vibration of atoms in the solid molecules will be more intense, which will generate greater Joule heat. For ions, when the temperature increases, it will accelerate the molecule movements, reduces the viscosity, which is conductive to reduce the energy loss caused by the ion movement. Studies have shown that ion movement is affected by temperature more significantly, indicating that when the temperature is increased, it is beneficial to reduce the energy loss of the super capacitor.
Compared with other electrochemical energy storage technologies, the ohmic polarization has less effect on supercapacitors. Since the typical application scenario of supercapacitors is high power, this application scenario needs to pay attention to two aspects. On the one hand, the ohmic polarization of supercapacitors will cause significant voltage changes, which will affect efficiency. Since the most significant feature of supercapacitors is the higher efficiency, the ohmic polarization can be used as an important indicator to judge the performance of the product; on the other hand, during the use process, it is necessary to avoid the danger caused by excessive temperature, and good thermal management is required in the product design.
High self-discharge is one of the main disadvantages of supercapacitors, which greatly limits the application of supercapacitors. In practice, the product energy retention time is relatively short, and some researchers have found that the capacity loss rate is up to 36% after 2 hours of storage. The capacity loss of the supercapacitor is mainly caused by the leakage point flow formed by the ions in the supercapacitor through the electrolyte membrane. The self-discharge rate of the supercapacitor is linearly related to the storage time. The researchers reduced the self-discharge performance of the supercapacitor through electrode coating, but sacrificed the energy density of the supercapacitor.
If you are interested in more details about relative products, please feel free to contact us, you can send a email to info@kamcap.com or dial at +86-18640666860.

